Neuronal circuits in the fovea
Context
In birds, the perception of moving objects – their speed, trajectory and direction – in a complex environment guarantees their survival. Little is known about neural circuits involved in high-frequency and high-resolution vision.
In the human fovea and, more generally in that of primates, the so-called midget pathway, consisting of a cone connected via one or two bipolar cells (BC) to one or two ganglion cells (GC), is the dominant neuronal circuit. This circuit is ideal for high spatial acuity but remains poorly suited to the perception of movement. The fovea of primates is viewed as being relatively inefficient in the processing of high frequency visual signals. Low spatio-temporal foveal acuity is the main reason why we perceive moving images rather than a sequence of separate images when watching a movie in the cinema or a video on our smartphone.


The high spatio-temporal perception of modern birds (Neoaves) suggests that their retinas and brain regions involved in vision are endowed with remarkable properties.
Behavioral experiments aimed at estimating temporal visual acuity have shown that some categories of birds can resolve alternating light-dark cycles at up to 145 Hz, which is 50 Hz over the highest frequency shown in any other vertebrate (Bostrom et al., 2016; Potier et al., 2020), suggesting motion perception in high resolution.
These observations lead to the question whether birds have a dedicated neural circuit in their fovea responsible for both high acuity and tracking fast moving objects (e.g., insects) ?
Approach
Two parallel pathways for visual signal transmission could exist side-by-side in the bird fovea :
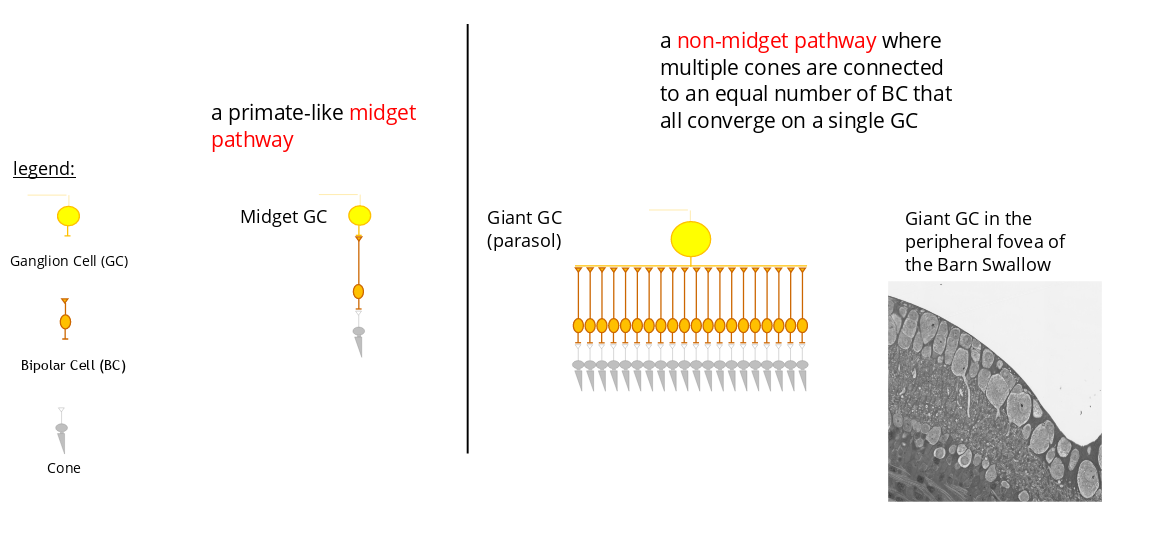
Midget et non-midget pathway in high acuity area of the retina
Experimental evidence in support of the presence of the midget et non-midget pathway in bird fovea are presented in Rodrigues and al. (2022) : https://www.biorxiv.org/content/10.1101/2022.10.29.514341v1.
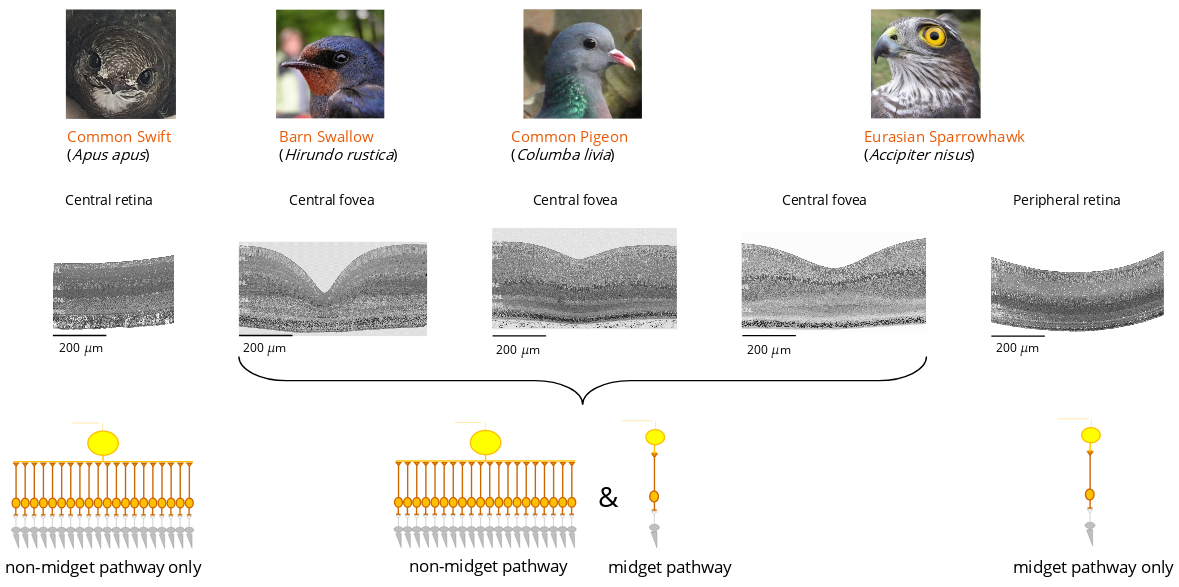
The presence of a non-midget pathway in the fovea would provide a wide range of sensitivity to rapid variations in light stimulation. It could prove to be essential for motion perception in high resolution.
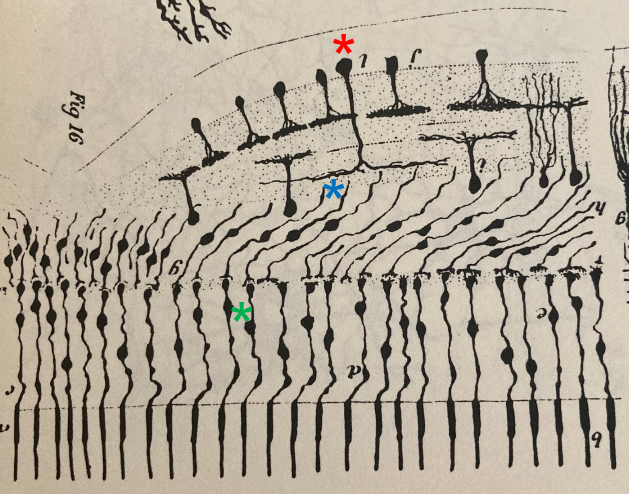
Santiago Ramon y Cajal (1852-1934) – the founding father of Neuroscience – had already anticipated the presence of a non-midget pathway in the fovea of birds. A drawing by his hand beautifully illustrates how the dendritic tree of a parasol GC (*) forms connections with several BCs (*), each of which is connected to a single cone (*)
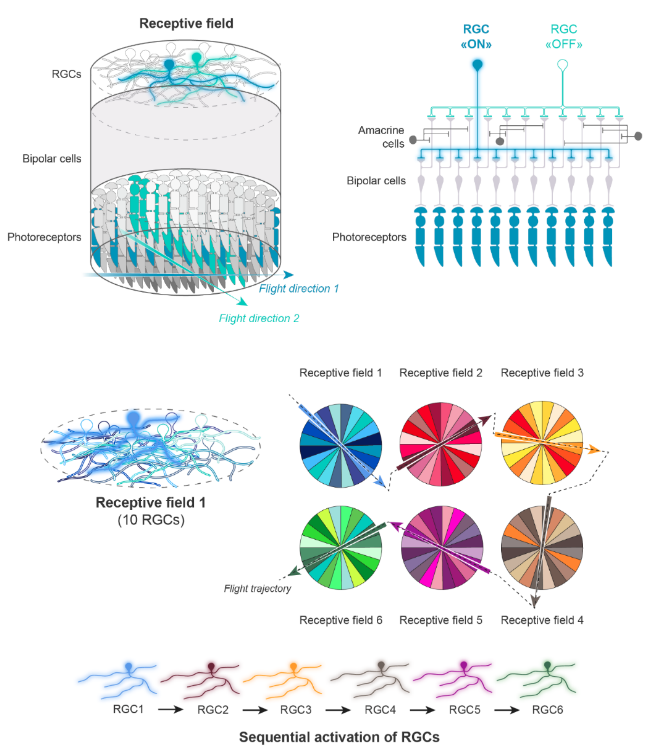
How could a neuronal circuit for high temporal acuity look like ?
In this model, we surmise that several RGCs (ten, in our example) have overlapping dendritic trees that define a single receptive field. In the example shown here, one RGC among ten (e.g., the light blue one in the two upper panels and in the receptive field 1) is activated (“ON”) by a moving object (e.g., a flying insect), because the blue photoreceptors that activate this RGC form an elongated excitatory domain that has the same orientation than the flickering light bar produced by the insect flying through the receptive field 1. The other nine RGCs (e.g., the turquoise one in the two upper panels and in the receptive field 1) remain “OFF” because they are connected, via BCs, to photoreceptors with excitatory domains that have different axes of orientation. The inhibitory effect of ACs on BCs like the ones depicted in the upper right panel can help keep these RGCs off. By moving (e.g., flying) across many receptive fields (only six are presented here), the object (e.g., insect) sequentially activates the RGCs having their axes of orientation aligned with the trajectory.
Identify and visualize the neurons of the fovea
In collaboration with the laboratory of Dr. Jean Livet :
https://www.institut-vision.org/fr/neurogenese-et-developpement-des-circuits-neuronaux.html :
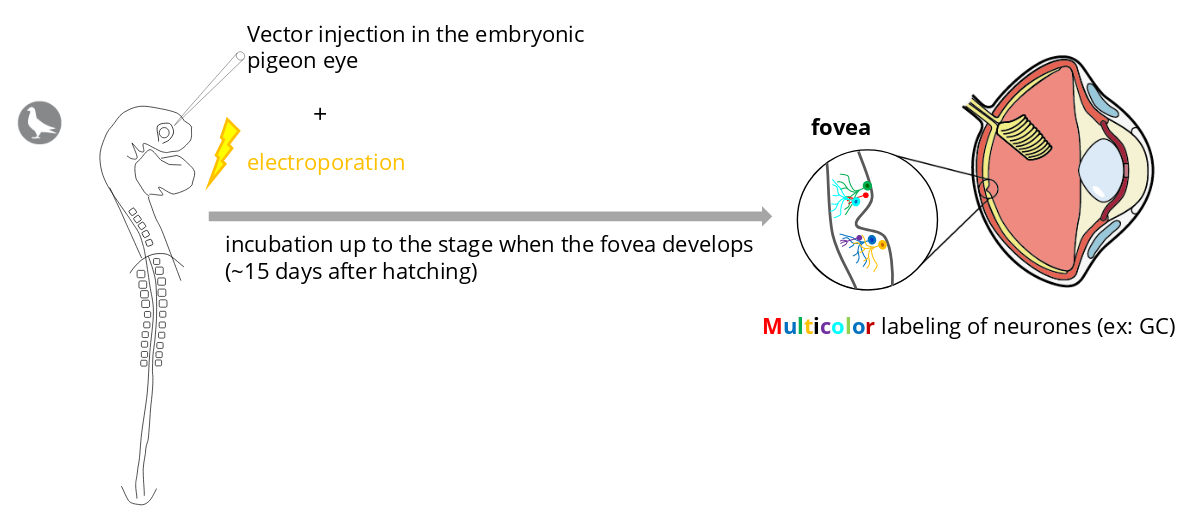
This experience should help us answer the following questions :
- Does midget and non-midget GCs are in the fovea ? If yes, in what proportions ?
- Does the fluorescent labeling of GCs provide information about the organization of dendritic trees within the fovea ?
- Do the dendritic trees of several GC overlap ?
Credits
This project is made possible thanks to a close collaboration with François Jardin, pigeon fancier and breeder :

